Understanding why evidence from randomised clinical trials may not be retrieved from Medline: comparison of indexed and non-indexed records. Making literature searches easier: a rapid and sensitive search filter for retrieving randomized controlled trials from PubMed. A simplified search strategy for identifying randomised controlled trials for systematic reviews of health care interventions: a comparison with more exhaustive strategies. Cochrane Handbook for Systematic Reviews of Interventions (2009) Higgins, J.P.T and Green, S., eds. Optimal search strategies for retrieving scientifically strong studies of treatment from Medline: analytical survey. Haynes RB, McKibbon KA, Wilczynski NL, Walter SD, Werre SR Hedges Team. Retrieving randomized controlled trials from medline: a comparison of 38 published search filters. McKibbon KA, Wilczynski NL, Haynes RB Hedges Team. It's been observed that many RCTs are now missed by the RCT publication type. However, the Cochrane Collaboration ceased tagging MEDLINE records in 2006. Used as limits or their underlying controlled vocabulary terms, they've been reported to produce extremely high precision (94%!) with only a small sacrifice in sensitivity. It is:įor precise limiting when you're pressed for time, you can use the randomized controlled trial limits in either PubMed or Embase. 'randomized controlled trial'/exp OR 'controlled clinical trial'/exp OR randomized:ti,ab OR placebo:ti,ab OR 'drug therapy':lnk OR randomly:ti,ab OR trial:ti,ab OR groups:ti,abĪs an alternative, Royle and Waugh published 'simplified' strategy that is nearly as effective as the foregoing methods (Sensitivity, 97% precision, 29% ), induces little or no bias, and is easy to transfer to interfaces other than PubMed. "randomized controlled trial" OR "controlled clinical trial" OR randomized OR placebo OR "drug therapy" OR randomly OR trial OR groups The "Highly Sensitive Search Strategy" has been used widely to retrieve randomized controlled trials and other types of evidence for systematic reviews. Section 6.4.11 of this Handbook includes strategies for limiting searches to controlled trials. The Cochrane Collaboration publishes an extensive handbook for systematic reviews. ('clinical':ti,ab AND 'trial':ti,ab) OR 'clinical trial'/exp OR random* OR 'drug therapy':lnk (clinical AND trial) OR "clinical trials as topic" OR "clinical trial" OR random* OR "random allocation" OR "therapeutic use" and provides high sensitivty (99%) at the expense of precision (10%). The NCBI's Clinical Queries for Therapy (sensitive) is based on work of Haynes, et al. One of the following filters should be adequate for most investigations. All rights reserved.There are dozens of filters available for limiting searches to randomized controlled trials. We suggest that levalbuterol should not be used over albuterol for acute asthma.Ĭopyright © 2012 Elsevier Ltd. Levalbuterol was not superior to albuterol regarding efficacy and safety in subjects with acute asthma. A few assumptions and some calculated values were used for meta-analysis. There were no significant differences in side effects between groups.ĭata were not available for two probable eligible trials. 
The results of the post hoc subgroup analyses that excluded the 2 studies from Mexico essentially confirmed the findings of the entire available data set. Respiratory rate, oxygen saturation, and percentage change in FEV1 and clinical asthma score were not significantly different between the groups with mean difference (95% CI) of 0.35 (-0.81, 1.51), -0.29 (-0.68, 0.10), -28.3 (-59.95, 3.33) and -1.01 (-5.30, 3.28) respectively. In the post hoc meta-regression analysis, the overall attrition rate and between-group attrition difference did not mediate any of the outcomes (eTable 6 and eTable 7 in the Supplement). Seven trials including a total of 1625 participants fulfilled the eligibility criteria. Meta-analyses were performed using Review Manager Software. 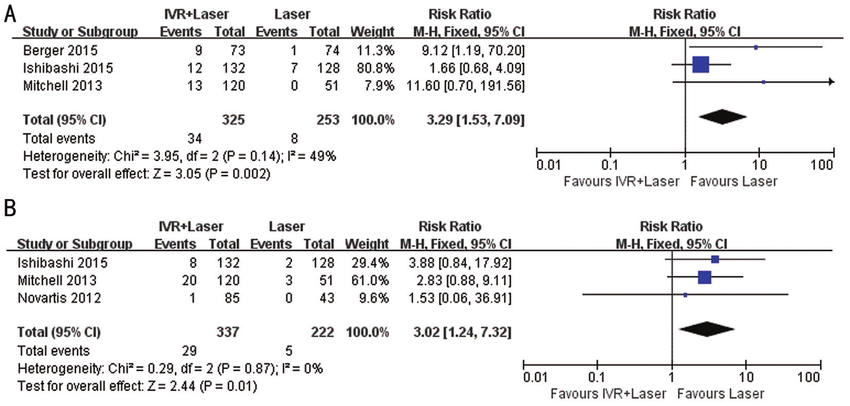
DATA EXTRACTION AND RESULT SYNTHESIS: Two authors extracted data independently. TRIAL ELIGIBILITY CRITERIA, PARTICIPANTS, AND INTERVENTIONS: Randomized control trials comparing levalbuterol versus albuterol for acute asthma in all age groups. To evaluate efficacy and safety of levalbuterol versus albuterol in acute asthma. Levalbuterol is therapeutically active component of albuterol whereas (S)-albuterol is considered inert with some unwanted effects. The Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement, published in 2009, was designed to help systematic reviewers transparently report why the review was done, what the authors did, and what they found. Conventional albuterol is a racemic mixture of (S)-albuterol and (R)-albuterol (levalbuterol).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
